|
A historical overview of the global steelmaking against recent developments and challenges of the novel technology is presented, and the fundamental mechanisms of iron oxide reduction to iron and alternative iron feedstocks are discussed. The present minireview discusses the progress on the electrochemical reduction of iron oxides in alkaline media as a green steelmaking route. Significant advantages of this technology include the absence of CO 2 emissions, non-polluting by-products such as hydrogen and oxygen gases, lower temperature against the conventional approach (∼100☌ versus 2000☌) and lower electric energy consumption, where around 6 GJ per ton of iron manufactured can be spared. In this scope, the electrochemical reduction or electrolysis of iron oxides into metallic iron in alkaline media arises as a promising alternative technology for ironmaking. Consequently, the steel sector is responsible for a large amount of CO 2 emissions, accounting for up to 9% of the CO 2 worldwide emissions. Traditional steelmaking industry still operates by the carbothermic reduction of iron ores for steel production. With a net zero carbon emissions target, European policies are expected to be accomplished before 2050. Steelmaking industries have been facing strict decarbonization guidelines. 2CIEPQPF, Department of Chemical Engineering, Pólo II-Pinhal de Marrocos, University of Coimbra, Coimbra, Portugal.1Department of Materials and Ceramic Engineering, CICECO-Aveiro Institute of Materials, University of Aveiro, Aveiro, Portugal.But there are several ways to prevent the rusting of iron.Daniela V. In this, iron reacts with oxygen and moisture present in the atmosphere. Rusting of iron is also an example of redox reactions. In redox reactions, red means reduction and ox means oxidation. The gaining of electrons is called Reduction, and the loss of electrons is called Oxidation. It has real-life uses in industry and manufacturing processes. The definition of a redox reaction occurs simultaneously. Oxidation and Reduction reactions play a very vital role. The methods mentioned above will help you to protect iron articles. They easily get rusted in the presence of oxygen and react with the moisture present in the environment. The articles of iron are very prone to rust. To avoid iron rusting, many methods can be applied. Iron hydroxides are also created from the natural interaction and the reaction between the iron cations and hydroxide ions.įe 3+ + 3OH – → Fe(OH) 3 How can the rusting of iron be prevented? The mentioned below are the acid-base reactions that occur between the iron cations and the water :. The iron oxidation state is somehow caused by the oxygen atom in the presence of water. The chemical reaction can be written as:. So, the iron atom instantly offers up electrons when revealed to oxygen. On the other hand, iron behaves as a reducing agent. The reaction that occurs in the formation of rust is mentioned below:. It takes place between oxygen and iron in an environment containing water as the air contains high moisture levels.Īll the reactions involved in rusting of iron The rust forms by the process of redox reaction. 
The construction of rust is a combination of iron oxides on the surface of iron objects or structures. The oxygen is also used to get combined with the metal iron, and it is a process of the reduction reaction, and the metal iron behaves as a reducing agent. It is an example of an oxidation reaction where oxygen acts as an oxidising agent. In the rusting process, iron is used to combine with oxygen in the presence of water. The rusting of iron is an example of a redox or oxidation-reduction reaction. Note that reduction potential is negative.ĢH+ + 2e- → H 2. Then simply, add concentration and pressure.  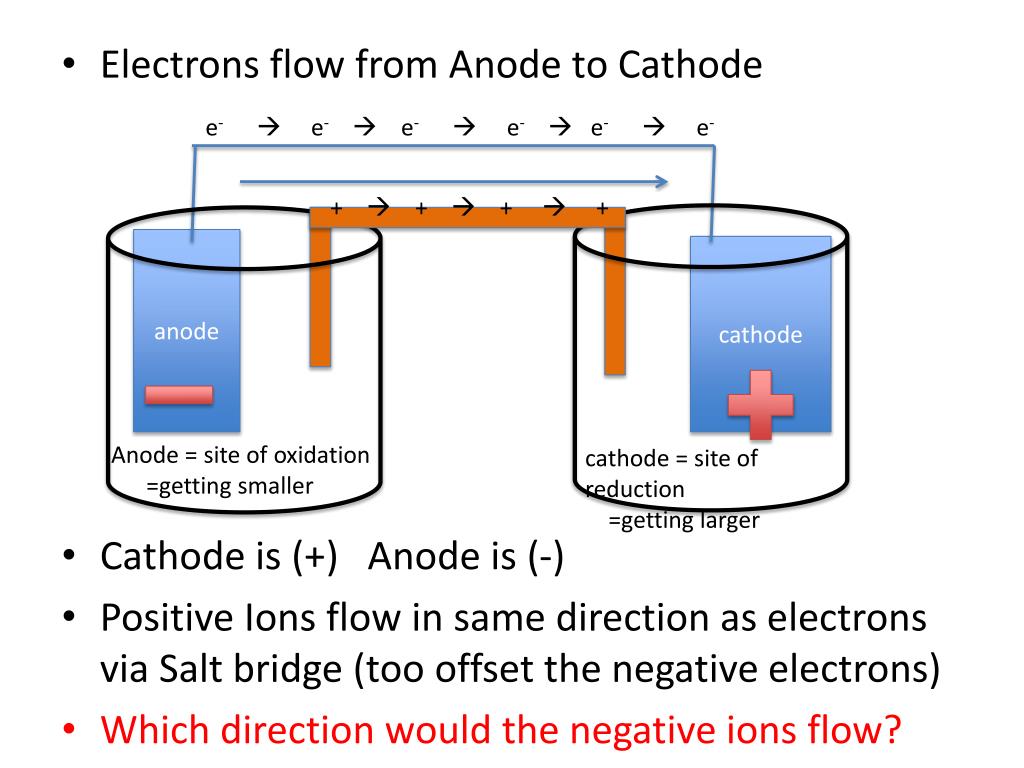
Remember:- To solve this equation, simply write the first half-reaction of reduction for hydrogen. The reduction potential of a hydrogen half-cell will be negative if: Standard cathode (reduction) Half-reaction
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
